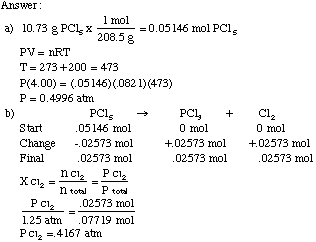
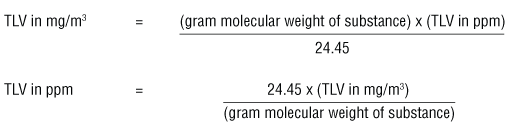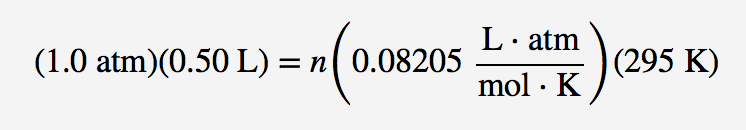SOLVED: Convert 798 mm Hg to atm. Show all work, including pre-rounded answers from the calculator and all units. A) 2.1 atm B) 1.05 atm C) 6.06x105 atm D) 0.952 atm

Ideal Gas Law Calculator (Pressure–Volume–Temperature–Amount) • Thermodynamics — Heat • Online Unit Converters

6.3: Combining the Gas Laws: The Ideal Gas Equation and the General Gas Equation - Chemistry LibreTexts

Gas Pressure Unit Conversions - torr to atm, psi to atm, atm to mm Hg, kpa to mm Hg, psi to torr - YouTube

Two moles of an ideal gas at 2 atm and 27^(@)C is compressed isothermally to half of its volume by external pressure of 4 atm. The work doen is

If I have 21 moles of gas held at a pressure of 3800 torr and a temperature of 627°C what is the volume of the gas? | Socratic