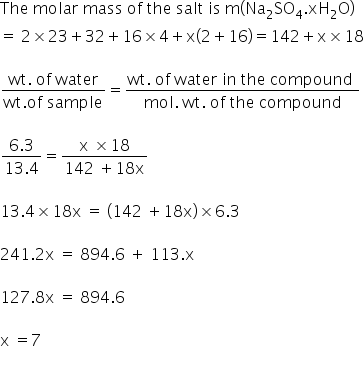Calculate the percentage of water of crystallization in CuSO4.5H2O (H = 1, O = 16, S = 32, Cu = 64) .
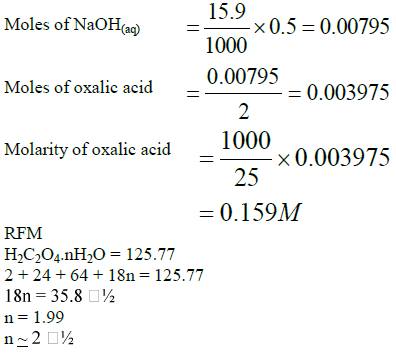
Calculate the number of molecules of water of crystallization in oxalic acid crystals, H2C2O4.nH2O given that 5g of the crystals were made upto 250cm³ of this...

SOLVED: Given the following set of data Mass of hydrated salt=1.14 g Mass of water = 0.50g Molar mass of anhydrous salt= 231 g/mol Molar mass of water= 18 g/mol Calculate water

Calculate the number of molecules of water of crystallization in copper sulphate crystals, if 10g of - Brainly.in

Water of Crystallisation Explain the terms: anhydrous, hydrated; and water of crystallisation. Calculate the formula of a hydrated salt using. - ppt download