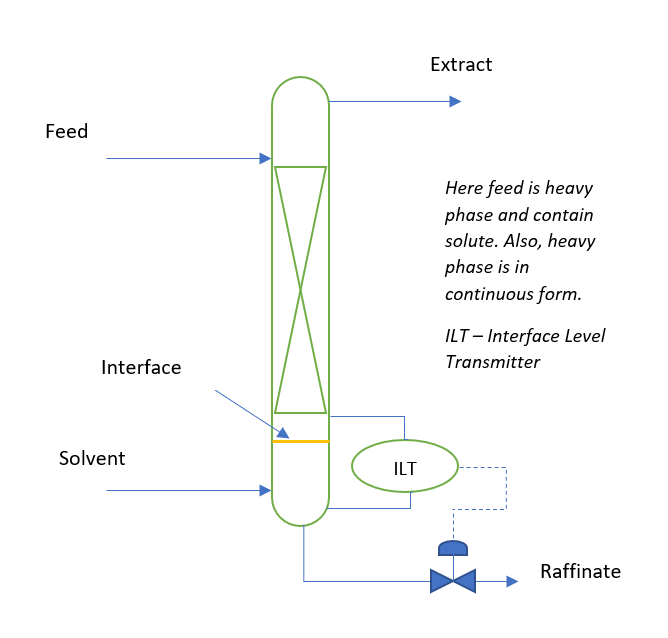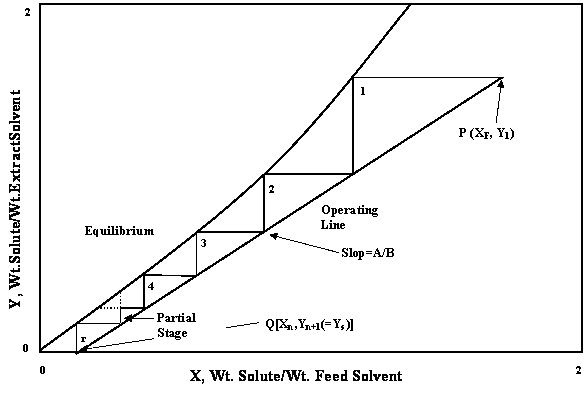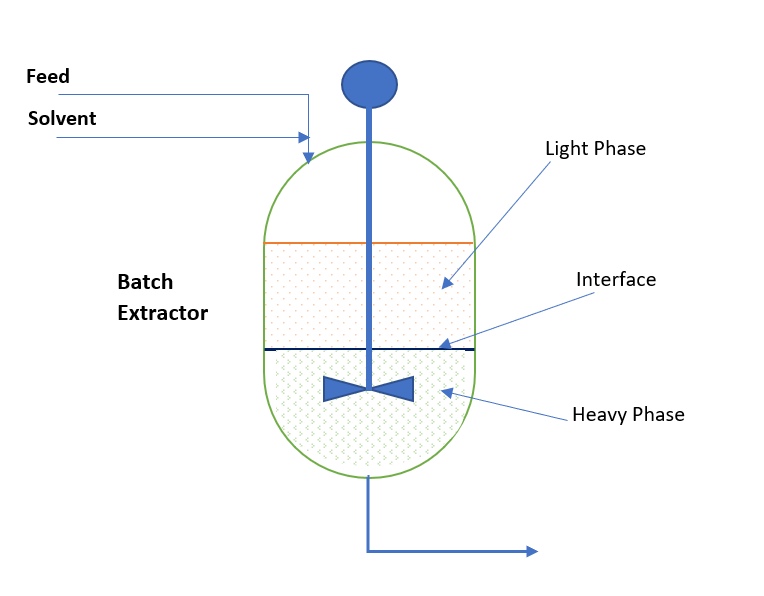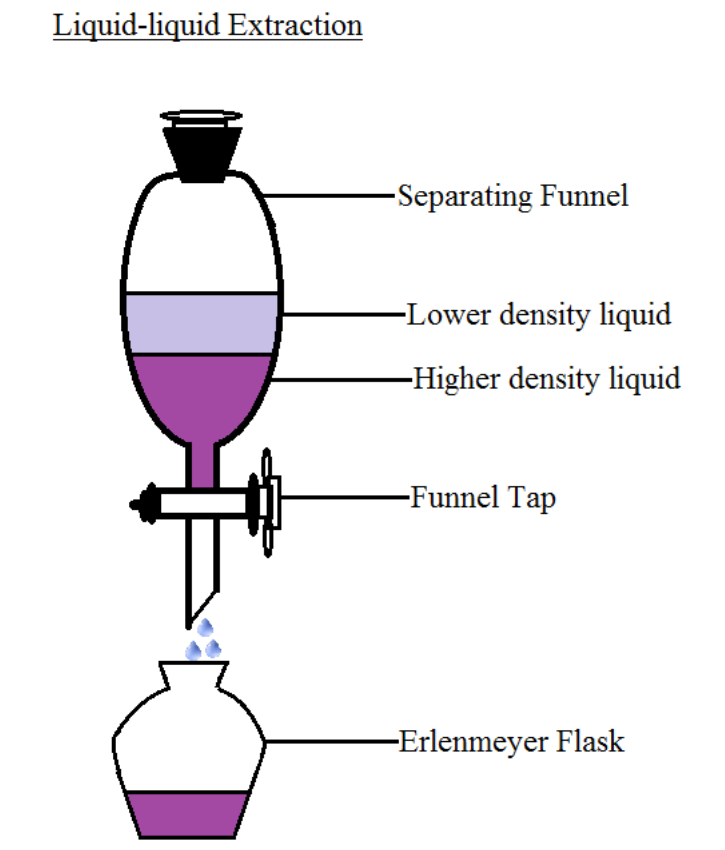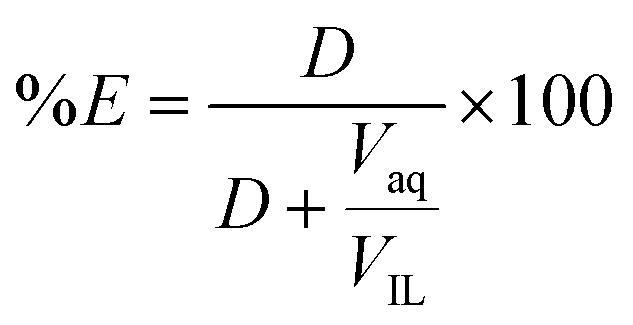
Liquid–liquid extraction of europium( iii ) and other trivalent rare-earth ions using a non-fluorinated functionalized ionic liquid - Dalton Transactions (RSC Publishing) DOI:10.1039/C3DT52285G

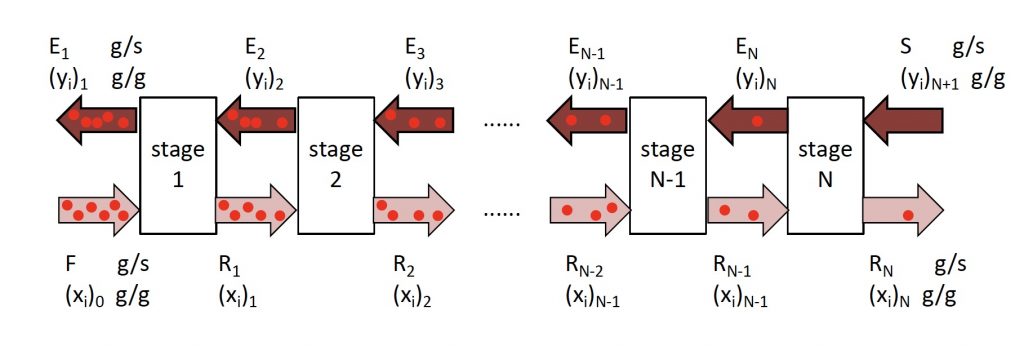
Stepwise equation assembly in phytoextraction (solid liquid extraction). | Download Scientific Diagram

SOLVED: Calculate the fraction of solute extracted (1-q) for a liquid-liquid extraction performed with the following parameters: Vsample 100 mL; Vorganic 100 mL; Partition coefficient (K) = 5 100 0.167 0.123 Correct

SOLVED: Caffeine from tea can be extracted by using a liquid-liquid extraction technique If 125 mL of tea solution contains 0.25 M of caffeine is shaken with 75 mL of dichloromethane 0.55